Specialist GVP Audit Services for Sponsors
The TGA’s Pharmacovigilance Inspection Program (PVIP) is now active for listed medicine sponsors, and inspection expectations have sharpened significantly. GxPVigilance provides independent GVP audit services designed specifically for sponsors operating in the registered, listed and OTC medicine space in Australia and New Zealand.
Led by Carl Bufe — Lead Quality Management System Auditor, QPPV, and ISO Lead Auditor (Medical Device and Artificial Intelligence) — every audit is grounded in your actual product risk profile, your regulatory obligations, and what TGA inspectors genuinely look for.
What Makes Us Different
Implementation Focus, Not Assessment-Only
Australian TGA Context Expertise
We are not translating generic international frameworks. We help develop systems that operate within your regulatory environment—aligning with Australian healthcare realities while remaining internationally compliant where necessary.
Capability Transfer, Not Dependency
You own the capability when we’re done—not just a list of corrective actions.
Patient Safety Non-Negotiable
Pharmacovigilance Services
At GxpVigilance, we help sponsors and MAHs run TGA-ready pharmacovigilance across Australia—built for clarity, control, and inspection readiness.
- PV system setup & governance (SOPs, SDEAs, oversight)
- AE intake/triage, case processing & regulatory reporting support
- Literature monitoring, signal oversight & safety documentation
- Local PV Contact Person / QPPV / A-PVCP support
Why GVP Auditing Matters
- Verify that adverse events and safety data are collected, assessed, and reported in line with required timelines.
- Confirm that case-management and signal-detection procedures are traceable, effective, and risk-proportionate.
- Ensure governance documents (SOPs, risk-management plans, signal trackers) remain current, auditable, and aligned with your obligations.
- Define local roles clearly (such as the Qualified Person for Pharmacovigilance in Australia) and support them with documented processes.
Comprehensive GVP Audit Scope
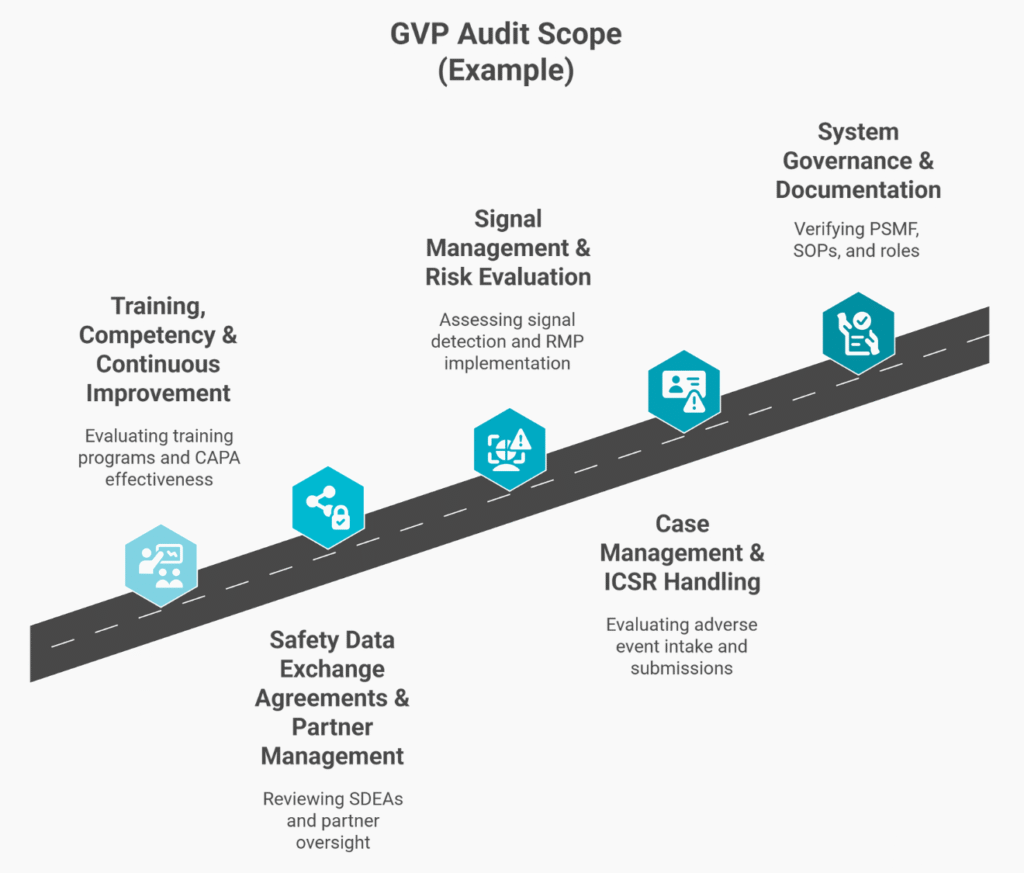
Our Approach
- Phase 1 — Initiation & Planning: Define purpose, scope, criteria, resources, and responsibilities; align objectives and timelines.
- Phase 2 — Engagement & Preparation: Confirm authority, communications, confidentiality, and logistics; draft a risk-based audit plan and checklists.
- Phase 3 — Document & Evidence Review: Collect and screen policies, procedures, and records to understand the system design and controls.
- Phase 4 — Audit Execution (Virtual/On-Site): Conduct interviews, observe, and collect samples; verify implementation against criteria with objective, traceable evidence.
- Phase 5 — Evidence Evaluation & Findings: Analyse conformity, classify nonconformities by risk/impact, and substantiate with objective evidence.
- Phase 6 — Reporting & Communication: Deliver a clear report of scope, evidence, findings, and conclusions; agree on corrective action priorities.
- Phase 7 — Follow-Up & Closure: Verify CAPA implementation and effectiveness; capture lessons learned and formally close the audit.
Who We Serve
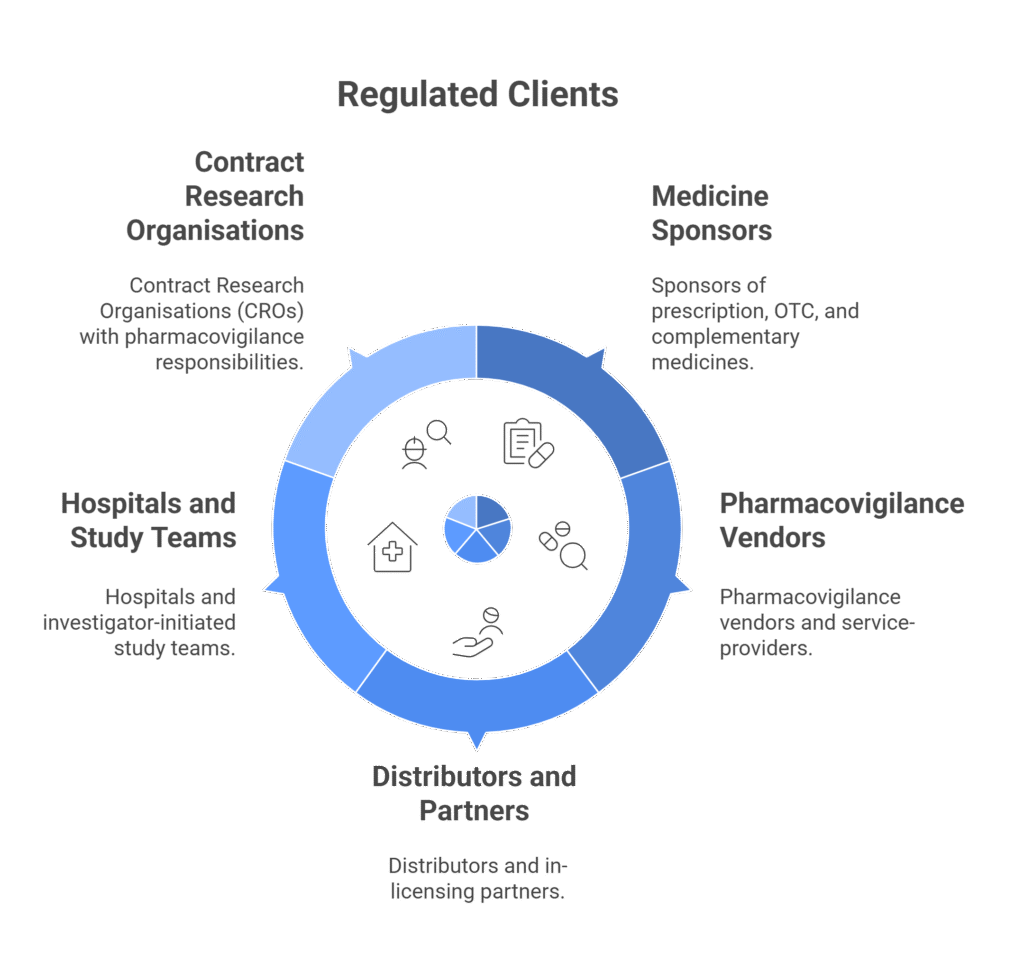
Why Choose GxP Vigilance
- Experienced Auditors: Our auditors hold decades of expertise in pharmacovigilance and regulatory affairs — they know what regulators expect, and they guide you accordingly.
- Local Expertise: We understand the TGA and New Zealand regulatory frameworks — and we align your systems with global best practice (including EU GVP guidance adopted in Australia).
- End-to-End Oversight: From planning through to CAPA closure and follow-up, we support you every step of the way.
- Human + AI Insight (Optional): For clients who adopt advanced tools, we incorporate AI-assisted analytics to enhance case tracking, literature review, and signal trend detection.
Common Questions and Answers
Do listed medicine sponsors need a pharmacovigilance audit?
Yes. The TGA’s pharmacovigilance obligations apply to all sponsors of listed and registered medicines on the ARTG. The TGA Pharmacovigilance Inspection Program (PVIP) audits listed medicine sponsors, and inspection findings are escalating. A GVP audit identifies gaps before a TGA inspector does.
What does an independent pharmacovigilance auditor actually do?
An independent PV auditor examines your pharmacovigilance system against GVP requirements and TGA expectations — reviewing SOPs, adverse event records, signal detection processes, QPPVA accountability, and SDEA documentation. Independence is critical: the auditor must have no involvement in operating the system they’re assessing. At GxPVigilance, we audit only — we do not run your PV system, which preserves objectivity.
How do I prepare for a TGA Pharmacovigilance Inspection Program (PVIP) inspection?
The most effective preparation is a mock TGA inspection conducted by an independent auditor familiar with PVIP criteria. This replicates the inspection experience — document requests, QPPVA interviews, adverse event record reviews — and produces findings you can remediate before the real inspection. We conduct mock PVIP inspections for listed and registered medicine sponsors across Australia.
What’s the difference between a GVP audit and a TGA inspection?
A TGA inspection is conducted by TGA officers under statutory authority. A GVP audit is an independent, voluntary assessment conducted ahead of inspection to identify gaps. Audits are your opportunity to find problems yourself. Inspections are the regulator’s opportunity to find them for you.
Can you audit both the pharmacovigilance and device vigilance systems at the same time?
Yes. For sponsors who hold both medicine ARTG entries and medical device registrations, GxPVigilance can conduct integrated PV and device vigilance audits, covering both the PVIP criteria and Medical Devices Vigilance Program (MDVP) expectations in a single engagement.
How often should we audit our pharmacovigilance system?
Most sponsors benefit from a risk-based GVP audit at least every 2–3 years, with more frequent targeted audits after major changes such as outsourcing PV, launching new high-risk products, or switching safety databases. A structured PV audit program also aligns with good quality management practice by ensuring that critical safety processes, vendors, and electronic systems are periodically reviewed for compliance and effectiveness.
What documents and systems are reviewed during a pharmacovigilance audit?
A PV audit typically assesses your pharmacovigilance system master file (PSMF), SOPs and work instructions, safety database configuration and validation evidence, training records, and key quality system elements such as deviations, CAPA, and change control. Auditors also sample individual case safety reports, aggregate reports, signal management outputs, and safety-related communications to verify that day-to-day practice matches your documented processes.
What are the most common findings in pharmacovigilance audits and inspections?
Frequent findings include outdated or incomplete SOPs, weak documentation of safety decisions, delayed or incomplete adverse event reporting, and insufficient training or oversight of vendors handling PV activities. Regulators and auditors also routinely identify deficiencies in validated electronic systems, such as missing audit trails, inadequate data reconciliation, and lack of evidence that safety databases were properly validated and periodically reviewed.
What is the QPPVA’s role in a GVP audit or TGA inspection?
The QPPVA (or Australian equivalent PV responsible person) is expected to demonstrate end-to-end oversight of the pharmacovigilance system, including awareness of key risks, critical vendors, and current CAPA. During audits and inspections, authorities often interview the QPPVA to confirm that responsibilities are clearly defined, that they have sufficient authority and resources, and that they are actively involved in monitoring system performance and compliance.
What happens after a pharmacovigilance audit — do you help with CAPA?
A high-quality GVP audit does more than list deficiencies: it prioritises findings by risk, supports root-cause analysis, and leads to targeted corrective and preventive actions (CAPA) with realistic timelines. While the sponsor owns implementation, an independent auditor can review CAPA plans and, where needed, perform follow-up audits to confirm that changes are effective before any TGA inspection.
Pharmacovigilance Services
At GxpVigilance, we help sponsors and MAHs run TGA-ready pharmacovigilance across Australia—built for clarity, control, and inspection readiness.
- PV system setup & governance (SOPs, SDEAs, oversight)
- AE intake/triage, case processing & regulatory reporting support
- Literature monitoring, signal oversight & safety documentation
- Local PV Contact Person / QPPV / A-PVCP support



